Chinese researchers made breakthrough in the field of electrode-electrolyte interface
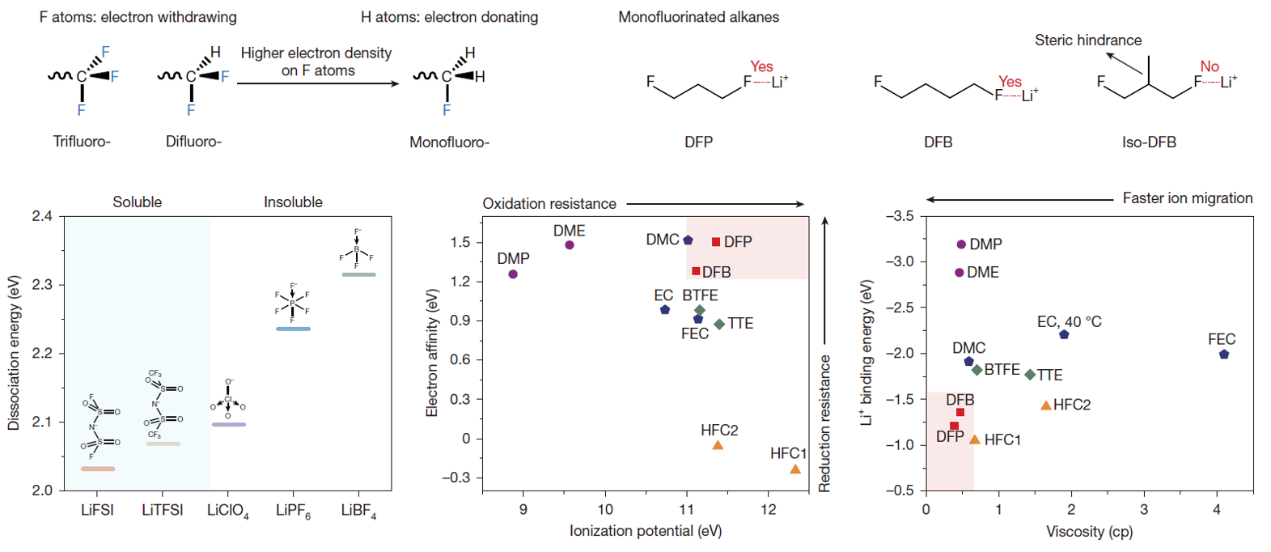
Figure. Design principles of F-coordinated hydrocarbon electrolytes
Supported by the National Natural Science Foundation of China (Grant Nos. 22393900, 22372083, 52201259, 22121005, 92472122), Prof. Zhao Qing and Prof. Chen Jun from the College of Chemistry, Nankai University, in collaboration with Prof. Li Yong from the Shanghai Institute of Space Power-Sources, have made progress in the field of electrolytes. The research entitled "Hydrofluorocarbon electrolytes for energy-dense and low-temperature batteries" has been published in Nature on February 26, 2026. The publication link: https://www.nature.com/articles/s41586-026-10210-6.
From volt batteries, lead-acid batteries, nickel-metal-hydride batteries to lithium (Li) batteries, every breakthrough in battery systems is inseparable from the innovation of electrolytes. For a long time, oxygen (O) atoms have been regarded as indispensable elements in electrolyte solvents. The ion-dipole interaction between charge carriers (Li+, Na+) and O provides the basis for ion dissociation and transport. However, this strong interaction also hinders the charge transfer process at the electrode-electrolyte interface, restricting the improvement of energy density and low-temperature performance of batteries. To address this issue, the aforementioned team designed and synthesized a series of hydrofluorocarbon solvent molecules. By regulating the electron density of fluorine (F) atoms and the steric hindrance of solvents molecules, the team achieved high dissolution of lithium salts in hydrofluorocarbon, and thus successfully replaced the traditional Li-O with Li-F coordination. By further optimizing the molecular structure, the team discovered the design principles of the electrolyte and the interfacial compatibility principles towards Li metal anode. Compared with the traditional electrolytes based on Li-O coordination, hydrofluorocarbon solvents are endowed with good wettability and high utilization rates, which can significantly reduce the amount of electrolyte used. Meanwhile, the Li-F coordination is much weaker than Li-O coordination, thus providing rapid interfacial charge transfer kinetics at low temperatures. Based on this design, the team achieved an ultra-high specific energy of 700 watt-hours per kilogram for room-temperature Li metal batteries that still maintained a high energy density of nearly 400 watt-hours per kilogram under an extremely cold environment of -50 ℃.
Contact Us

National Natural Science Foundation of China
Add: 83 Shuangqing Rd., Haidian District, Beijing, China
Postcode: 100085
Tel: 86-10-62327001
Fax: 86-10-62327004
E-mail: bic@nsfc.gov.cn
