Chinese Scholars Make Progress in Main Group Element Catalysis
With support from projects funded by the National Natural Science Foundation of China (Grant Nos. 22350004, 22301119, and 22271132), Professor Liu Leo Liu’s group at Southern University of Science and Technology has made progress in main group element catalysis. The research, entitled “Aluminium redox catalysis enables cyclotrimerization of alkynes”, was published in Nature on February 11, 2026. The paper is available at: https://www.nature.com/articles/s41586-025-09941-9.
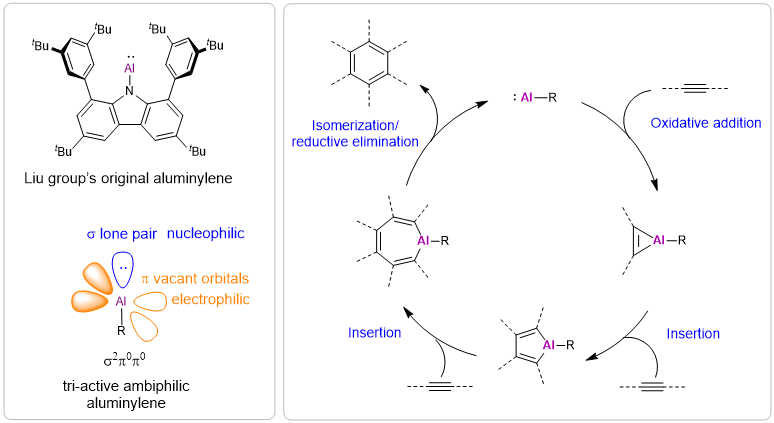
Figure. Aluminium redox catalysis enables cyclotrimerization of alkynes
Aluminium is the most abundant metallic element in the Earth’s crust (8.1%). As a representative third-period main group element, aluminium is essentially free from inert-pair effects and exhibits the lowest electronegativity (1.61) among p-block elements. Moreover, its valence electronic structure strongly biases it toward three-electron oxidation, rendering aluminium(III) compounds thermodynamically favored under a wide range of conditions. Since the first organoaluminium compound was synthesized in 1859, catalytic applications of aluminium(III) species have therefore been dominated by Lewis-acid paradigms, whereas an aluminium redox catalytic platform has remained elusive. Establishing aluminium redox catalysis would not only sharpen our mechanistic understanding of aluminium reactivity, but could also expand the conceptual repertoire of main group catalysis.
To address this long-standing limitation, the team leveraged an aluminylene—an aluminium(I) species that can be viewed as an aluminium analogue of carbenes—whose Lewis amphiphilicity combines nucleophilic and electrophilic responses within a single reactive centre. This electronic duality enables key elementary steps with alkynes, including oxidative addition and insertion at aluminium. Moreover, conformationally adaptive regulation by the nitrogen atom embedded in the ligand’s carbazole framework was used to fine-tune the coordination environment around aluminium, thereby promoting the isomerization and reductive elimination events required to close the catalytic loop. Collectively, this delivered a recyclable aluminium(I)/aluminium(III) redox cycle that mediates efficient cyclotrimerization of alkynes, outlining a general strategy for engaging main group elements in redox catalysis and extending the scope of aluminium chemistry beyond classical Lewis-acid activation.
Contact Us

National Natural Science Foundation of China
Add: 83 Shuangqing Rd., Haidian District, Beijing, China
Postcode: 100085
Tel: 86-10-62327001
Fax: 86-10-62327004
E-mail: bic@nsfc.gov.cn
