Chinese scholars achieve significant advances in understanding the role of endogenous retroviruses in human early embryogenesis
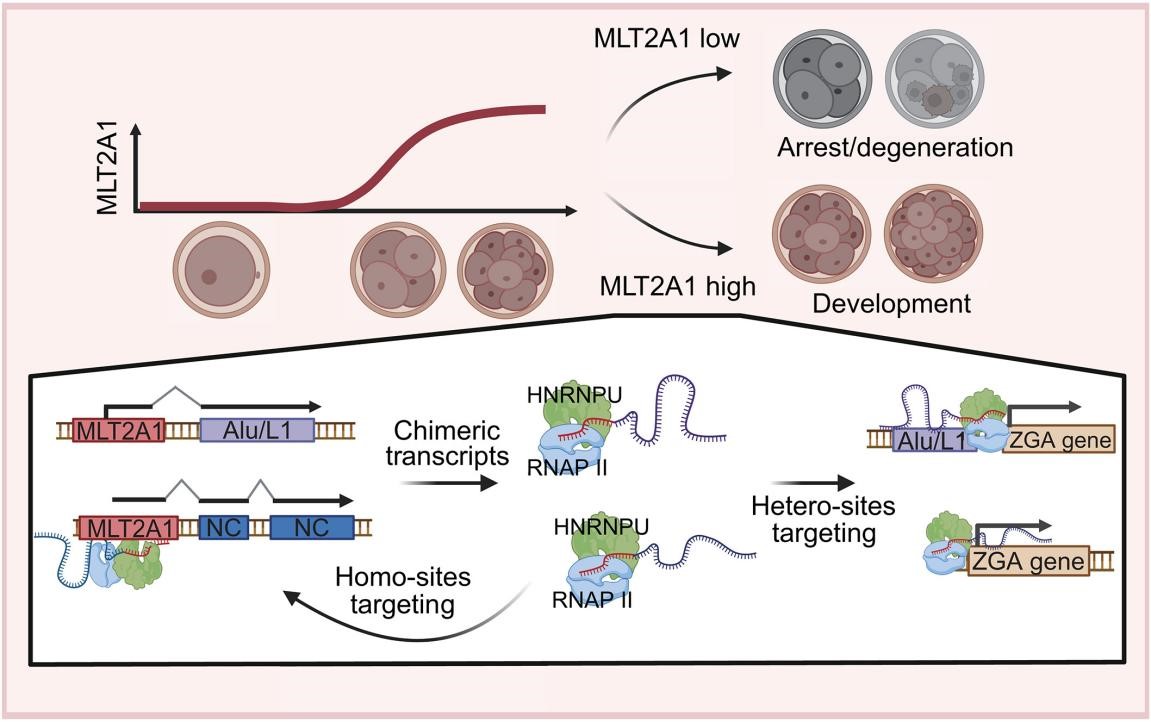
Figure. MLT2A1 enhances human ZGA by synthesizing heterologous chimeric RNA
Under the support of the National Natural Science Foundation of China (Grant Nos.: 32470783, 82394424, 32488101), the team led by Hongqing Liang and Dan Zhang (Zhejiang University), in collaboration with Shaorong Gao (Tongji University) and Wanlu Liu (Zhejiang University–University of Edinburgh Joint Institute), has achieved significant advances in understanding the role of endogenous retroviruses in human early embryogenesis. The work, entitled “Endogenous retroviruses synthesize heterologous chimeric RNAs to reinforce human early embryo development”, was published online in the journal Science on January 22, 2026. Paper link: https://www.science.org/doi/10.1126/science.adv5257.
Human embryonic development frequently arrests at critical stages between fertilization and implantation, contributing to infertility and pregnancy loss. Zygotic genome activation (ZGA) is a pivotal event during which the embryo initiates transcription from its own genome, and its failure is a major cause of early embryonic arrest. While studies in mammalian models have identified essential protein-coding genes within the ZGA network, the mechanisms underlying human ZGA failure remain poorly understood. Endogenous retroviruses (ERVs), a class of retrotransposable elements (retro-TEs) with numerous homologous copies dispersed throughout the genome, are highly activated during ZGA and constitute a substantial portion of the species-specific transcriptome. Nonetheless, their exact role in ZGA remains unclear.
To address these challenges, the research team sequenced human embryos arrested at the 8-cell ZGA stage and observed significant downregulation of the ERV subfamily MLT2A1. Knockdown of MLT2A1 in embryos reduced the 8-cell development rate from 47% to 16% and markedly decreased expression of key ZGA genes. Using Nanopore long-read sequencing, the team discovered that multiple copies of MLT2A1 form chimeric transcripts with downstream sequences. Over 70% of these chimeric RNAs fused with other retro-TEs (e.g., LINE1 and Alu), greatly expanding the sequence complexity of MLT2A1 transcripts. Biochemical analyses further revealed that MLT2A1 RNAs employ their diverse 3′ fusion sequences to broadly bind ZGA genes and their regulatory regions, while their conserved 5′ sequences specifically bind the nuclear protein HNRNPU, recruiting RNA polymerase II to activate nearby ZGA genes. Importantly, the researchers demonstrated that distinct MLT2A1 copies synergistically activate one another, forming a self-amplifying cooperative network that globally orchestrates ZGA gene induction (Figure).
This study elucidates how endogenous retrovirus transcripts regulate human early embryogenesis through novel mechanisms, including chimeric RNA formation, expanded genome targeting, and an interlocking subfamily network. These findings not only deepen our understanding of human embryonic development from its earliest stages but also propose retro-TE-derived RNAs as potential surrogate markers for assessing IVF embryo quality and monitoring successful ZGA progression.
Contact Us

National Natural Science Foundation of China
Add: 83 Shuangqing Rd., Haidian District, Beijing, China
Postcode: 100085
Tel: 86-10-62327001
Fax: 86-10-62327004
E-mail: bic@nsfc.gov.cn
