Chinese scholars have made progress in developing new types of pericyclic reactions
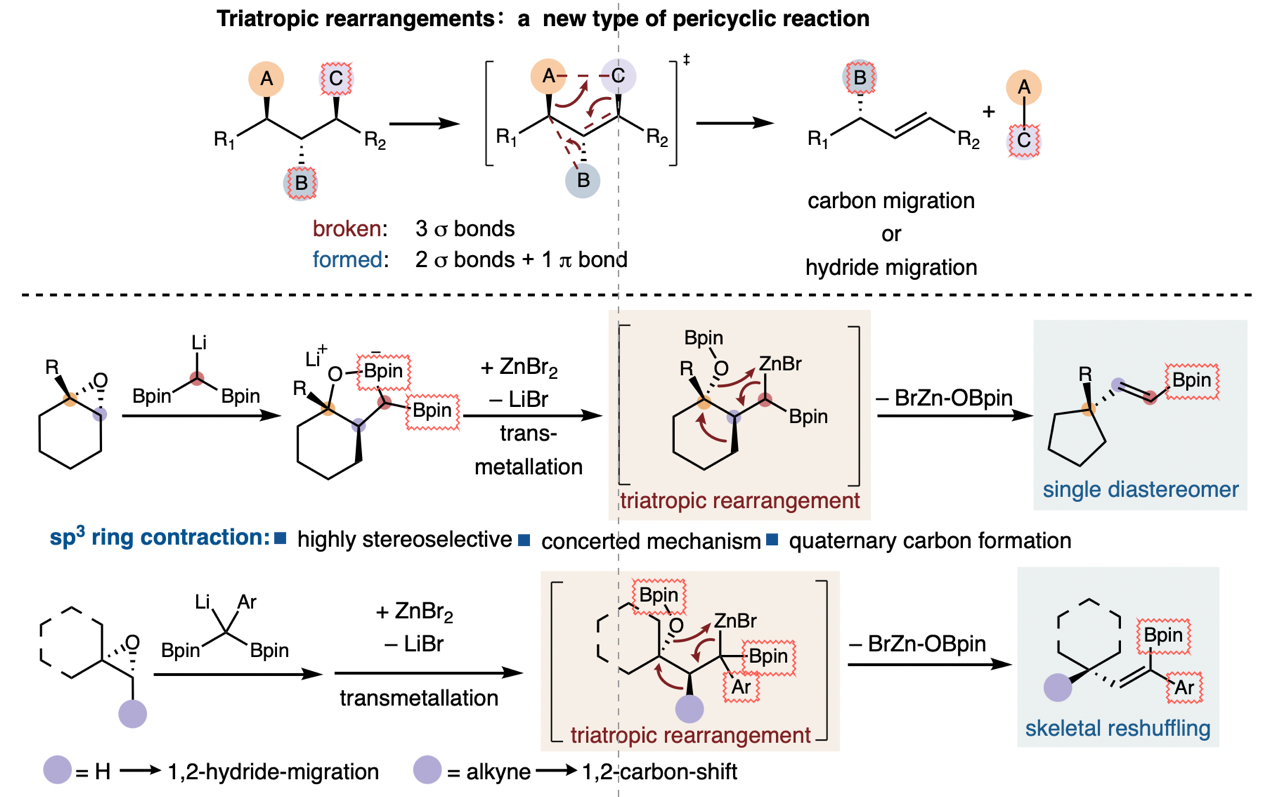
Figure Leveraging triatropic rearrangements for stereoselective skeletal reshuffling
Supported by the National Natural Science Foundation of China (Grant No. 22301123), the research groups led by Zhe Dong and Peiyuan Yu from Southern University of Science and Technology collaborated to achieve important progress in the field of pericyclic reaction research. The related findings entitled “Leveraging Triatropic Rearrangements for Stereoselective Skeletal Reshuffling” were published online in the journal Science on January 8, 2026. Link to the paper: https://www.science.org/doi/10.1126/science.adw3340.
Constrained by cyclic transition states, pericyclic reactions usually exhibit favorable stereoselectivity, enabling the rapid and efficient construction of complex molecules. They have greatly enriched the synthetic toolkit of organic chemistry and vigorously advanced the development of frontier orbital theory. The utilization of pericyclic reactions for the ring-system modification of cyclic molecules is of great significance, yet it has long been limited by numerous challenges such as low levels of chemoselectivity, regioselectivity and stereoselectivity.
To address these challenges, the research team developed an entirely new triatropic rearrangement reaction, which can cleave three σ bonds with high stereoselectivity in a single transition state while simultaneously forming two σ bonds and one π bond. This reaction provides a novel strategy for the cleavage and recombination of carbon-carbon bonds in saturated systems. This pericyclic reaction enables the stereospecific deoxygenative carbon-carbon bond formation of chiral epoxides, and also allows either the formal 1,2-hydrogen migration or 1,2-alkynyl migration of exocyclic and alkenes/acyclic alkenes. The core mechanism is as follows: a gem-diborylmethanide is employed as the nucleophile to ring-open the epoxy group and generate cyclic boronic esters with three well-defined contiguous stereocenters. After undergoing zinc bromide-mediated transmetalation, organozinc compounds are formed and participate in the triatropic rearrangement reaction, ultimately yielding alkenyl boronic ester products. Using this reaction, chiral cyclohexenes can be efficiently and highly selectively converted into chiral cyclopentanes. Moreover, by coupling with the Diels-Alder [4+2] cycloaddition reaction, chiral five-membered ring compounds can be rapidly prepared. This study fully demonstrates the great potential of triatropic rearrangement reactions, offering new methodologies for the development of novel reactions and the construction of complex pharmaceutical molecular skeletons.
Contact Us

National Natural Science Foundation of China
Add: 83 Shuangqing Rd., Haidian District, Beijing, China
Postcode: 100085
Tel: 86-10-62327001
Fax: 86-10-62327004
E-mail: bic@nsfc.gov.cn
