Chinese Scientists Made New Progress in Barocaloric Refrigeration
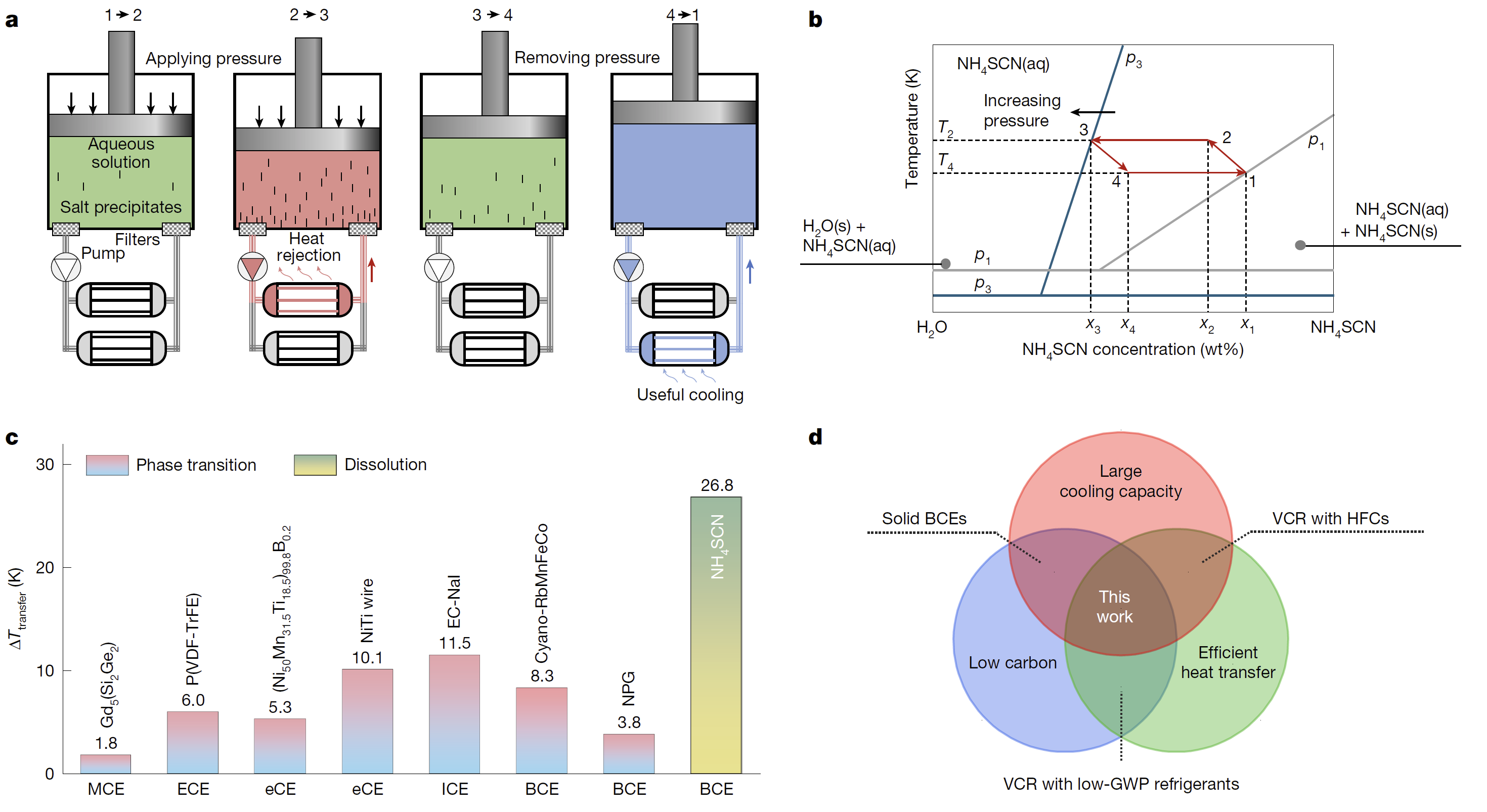
Figure: Principles and characteristics of extreme barocaloric effect at dissolution. a, Schematic diagram of the Carnot-like cooling cycle with four steps using the NH4SCN aqueous solution. b, Carnot-like cooling cycle illustrated on the phase diagram of the NH4SCN aqueous solution. c, Comparison of the maximum temperature drop of heat-transfer fluid (ΔTtransfer) of NH4SCN at dissolution with state-of-the-art caloric materials. d, Venn diagram comparing the refrigeration technologies and their merits.
Under the support of the National Natural Science Foundation of China's Young Scientists Fund (Tier A) (Grant No. 52425107) and others, the research team led by Prof. Li Bing from the Institute of Metal Research, Chinese Academy of Sciences, has made new progress in the field of barocaloric refrigeration. The research findings, titled "Extreme barocaloric effect at dissolution," were published in Nature on January 22, 2026 (Article link: https://www.nature.com/articles/s41586-025-10013-1).
Refrigeration technology is of vital importance for modern human civilization. In recent years, refrigeration technology based on the caloric effects of solids has received widespread attention for its potential to avoid greenhouse gas emissions. However, solid-state caloric refrigeration technology still faces a prominent inherent problem: unlike the mass and heat transfer of gaseous working fluids in vapor compression refrigeration technology, solids can only transfer heat through thermal conduction. Solid refrigerants must rely on fluids for indirect heat exchange with heat exchangers, and the presence of interfacial thermal resistance limits the system's refrigeration efficiency and high-power applications. Therefore, achieving a refrigeration technology that is compatible with low carbon emissions, high cooling capacity, and high heat exchange efficiency has become a key issue in the field of refrigeration materials.
Solid NH4SCN is highly soluble in water and its dissolution is strongly endothermic. For a near-saturated 60 wt% solution, dissolution at room temperature can be completed within 20 seconds, producing a temperature drop of up to 30 K. This temperature drop rapidly increases with rising environment temperature, reaching 54 K at 345 K. Using a custom-built instrument, the researchers measured in-situ temperature changes during pressure cycling were measured, achieving a temperature drop of 26.8 K under 600 MPa. This value is significantly superior to that of known solid-state caloric materials (as shown in the figure). Using in-situ synchrotron X-ray diffraction, Raman scattering, and microscopic observation, the team determined that the physical origin of the dissolution barocaloric effect is the significant reduction of solubility by pressure, and this process has advantages such as reversibility and rapid response. Based on the merits of high cooling capacity and fluid-mediated heat exchange of the dissolution barocaloric effect, a Carnot-like refrigeration cycle was designed. As shown in the figure, this cycle includes four stages: adiabatic pressurization leading to precipitation and temperature rise of the solution; maintaining pressure and circulating the solution to dissipate heat; stopping the circulation and adiabatic depressurization for self-cooling; and pumping the cooled solution to cool the heat source. Calculations show that the cooling capacity and efficiency of a single refrigeration cycle reach 67 J g-1 and 77%, respectively.
The advantages of high cooling capacity and fluid heat transfer of this technology make it promising for the heat management of high-power computing centers. At the same time, compared to the refrigeration based on phase transitions, this work utilizing dissolution enthalpy heat may inspire the discovery of new refrigeration approaches in the future.
Contact Us

National Natural Science Foundation of China
Add: 83 Shuangqing Rd., Haidian District, Beijing, China
Postcode: 100085
Tel: 86-10-62327001
Fax: 86-10-62327004
E-mail: bic@nsfc.gov.cn
