Chinese researchers make advances in the treatment of progressive multiple sclerosis
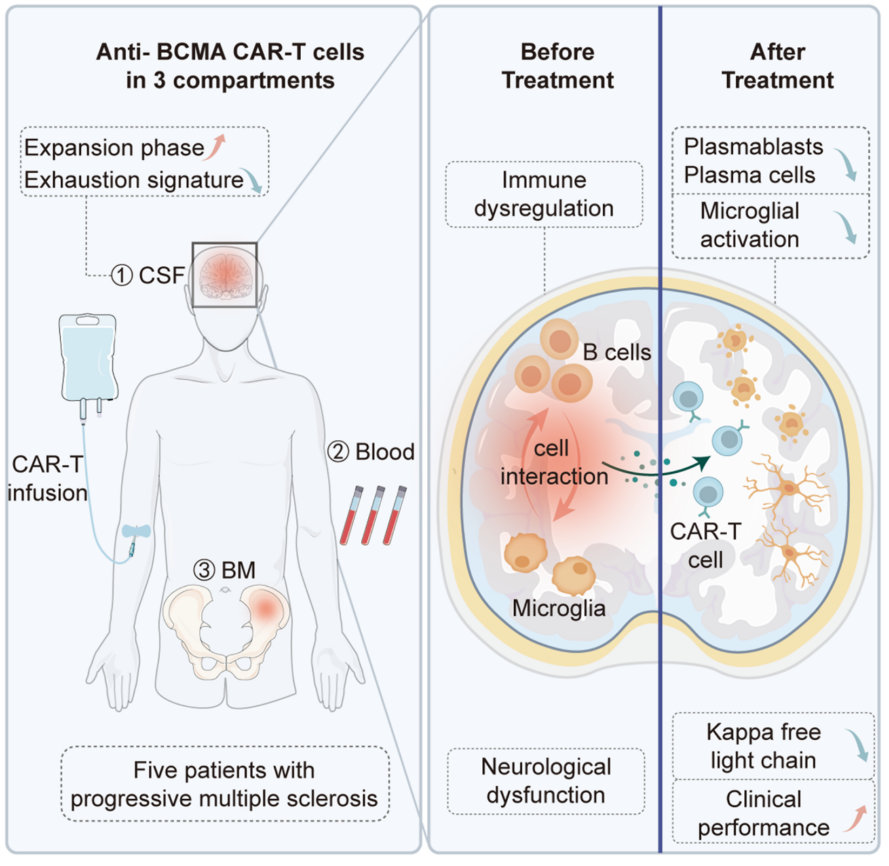
Figure. Mechanism of BCMA‑targeted CAR‑T Cell Therapy in Progressive Multiple Sclerosis
Supported by the National Natural Science Foundation of China (Grants 82371404, 82071380, 82471353, 82271341), a team led by Professors Wang Wei, Tian Dai‑Shi, and Qin Chuan from Tongji Hospital, Huazhong University of Science and Technology has made progress in treating progressive multiple sclerosis. Their study, entitled “Anti‑BCMA CAR‑T therapy in patients with progressive multiple sclerosis”, was published online in Cell on November 13, 2025 (https://doi.org/10.1016/j.cell.2025.09.020).
Progressive multiple sclerosis is a refractory central nervous system disorder and a leading cause of non-traumatic disability in young adults worldwide. It is marked by persistent neurodegeneration and irreversible disability accumulation, driven mainly by compartmentalized neuroinflammation. Conventional therapies often fail to adequately reach lesion sites, resulting in limited clinical benefit for most patients. B cells and their terminally differentiated plasma cells contribute to sustained autoimmune activity in the CNS by promoting antigen presentation, secreting pro-inflammatory factors, and forming ectopic lymphoid structures. Thus, targeted depletion of these cells is considered a key strategy to halt disease progression. However, current treatments cannot effectively eliminate long-lived plasma cells within the CNS, a major obstacle to clinical advances.
The team evaluated the feasibility and clinical efficacy of anti-BCMA chimeric antigen receptor T (CAR-T) cell therapy in patients with progressive multiple sclerosis. The treatment proved safe, with no serious adverse events reported. Importantly, CAR-T therapy cleared plasma cells from the central nervous system, reflected by significant decreases in cerebrospinal-fluid oligoclonal bands and κ-free light chains. Further analyses showed reduced microglial activation and downregulation of inflammatory pathways, providing a mechanistic explanation for the decrease in neuroinflammation. Moreover, CAR-T cells displayed distinctive in vivo distribution, sustaining expansion across blood, bone marrow, and the CNS while exhibiting a less exhausted phenotype and prolonged survival in the cerebrospinal fluid (Figure).
This study demonstrates the safety and clinical potential of anti-BCMA CAR-T therapy in progressive multiple sclerosis. By effectively removing pathogenic cells and potentially reshaping the immune microenvironment toward a sustained immune reset, it offers a promising new direction for treating this challenging disease.
Contact Us

National Natural Science Foundation of China
Add: 83 Shuangqing Rd., Haidian District, Beijing, China
Postcode: 100085
Tel: 86-10-62327001
Fax: 86-10-62327004
E-mail: bic@nsfc.gov.cn
